




REQORSA® by Genprex is a cutting-edge cancer treatment, delivering TUSC2 and NPRL2 genes via a non-viral lipid nanoparticle system. Designed to reverse glycolysis in cancer cells, it aims to restore tumor suppressor functions and enhance anti-cancer mechanisms. With promising results, REQORSA® represents a new frontier in cancer therapy.
"Genprex is excited by the early and promising patient responses to REQORSA® treatment, particularly as these patients represent some of the most difficult-to-treat lung cancer populations. There is a significant unmet medical need for patients afflicted with lung cancer, as nearly all patients' disease progresses following treatment, even when treated with today's most advanced targeted therapies and immunotherapies. This leaves patients with limited therapeutic options. Genprex is thrilled that the novel gene therapy treatment for lung cancer, REQORSA®, is demonstrating early evidence of efficacy with a favorable safety profile. The Company looks forward to continuing the evaluation of REQORSA® in lung cancer clinical trials while advancing efforts to bring new therapies to those battling cancer."
By Ryan Confer, President and Chief Executive Officer of Genprex.

In 2023, Genprex also received a third Fast Track Designation for REQORSA® Immunogene Therapy from the U.S. Food and Drug Administration, this time in combination with Tecentriq® for the treatment of small cell lung cancer. Additionally, REQORSA® was granted Orphan Drug Designation for the treatment of small cell lung cancer. Genprex believes these designations underscore and further validate the potential of REQORSA®. The Company’s accomplishments in 2023, which also include process improvements in manufacturing operations and securing new supplies of REQORSA®, set the foundation for a strong 2024.



Genprex, Inc. (NASDAQ:GNPX) investors are integral to our the Company’s mission of revolutionizing healthcare through groundbreaking gene therapies. With the support of stakeholders, Genprex is reshaping treatment possibilities for patients grappling with cancer and diabetes. Driven by a steadfast dedication to innovation and pioneering scientific discoveries, Genprex is propelling forward as a leading force in the biotechnology sector.
Building on Success: Genprex enters 2024 on a foundation of notable achievements in 2023. The completion of the Phase 1 segment of the Acclaim-1 trial, coupled with FDA Fast Track Designations and Orphan Drug Designations, underscores Genprex's commitment to innovation in cancer and diabetes therapies. These milestones signal the company's readiness to deliver impactful treatments and drive shareholder value.
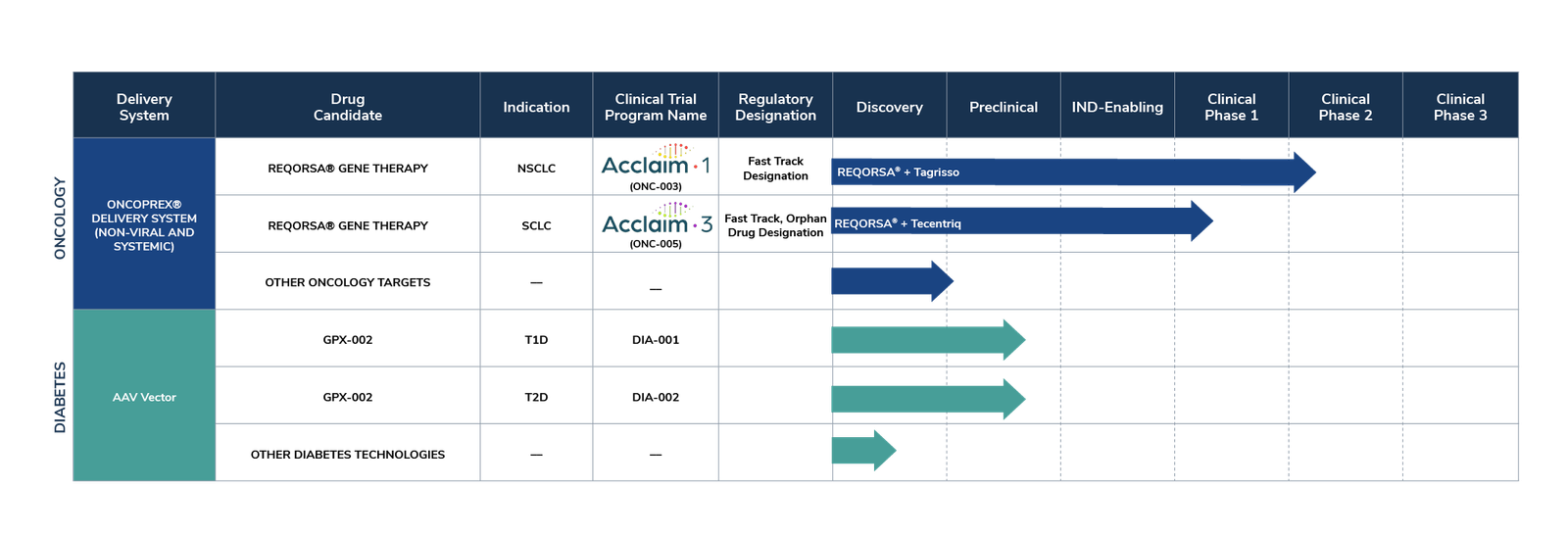
Oncology Innovation: Genprex's oncology gene therapy platform, anchored by the ONCOPREX® Nanoparticle Delivery System, continues to advance. With REQORSA® leading in two clinical trials targeting NSCLC and SCLC, and an expanding patent portfolio, Genprex seems to be positioned as leaders in cancer therapy innovation, promising long-term value for investors.
Clinical Milestones: In 2024, pivotal advancements are expected, including the Phase 2a expansion of Acclaim-1 and initiation of Acclaim-3 Phase 1. With encouraging Phase 1 Acclaim-1 results and ongoing collaborations driving preclinical data presentations, Genprex seems to be poised for clinical success and value creation.
Genprex's portfolio isn't limited to oncology; it extends to tackling the diabetes epidemic. The Company's gene therapy program for diabetes, centered around GPX-002, aims to restore insulin levels and revolutionize the lives of individuals impacted by diabetes.
Strategic Collaborations: Partnerships with institutions like the University of Pittsburgh strengthen Genprex’s R&D capabilities. Collaborations drive research advancements, enhancing the Company’s position as pioneers in gene therapy. Abstract submissions and data presentations at esteemed conferences underscore Genprex’s commitment to transparency and progress sharing.
Global Impact: With a growing global intellectual property portfolio, continuous collaborations, and strategic alliances, Genprex seems to be on the path to monumental expansion. Embark on this thrilling expedition with Genprex as the Company venture into new therapeutic areas, involve influential thought leaders, and pave the path for cutting-edge gene therapies in the 21st century.



"Genprex is excited to take this next step in the fight against lung cancers as the Company works to advance an innovative therapy that is believed to provide hope to patients suffering from ES-SCLC, an especially aggressive form of lung cancer with extremely limited treatment options. With a median progression-free survival (PFS) of 5.2 months, ES-SCLC has a particularly poor prognosis. Additionally, patients receiving Tecentriq as maintenance therapy have a median PFS of 2.6 months after the start of maintenance therapy. With such limited benefit from current treatments, Genprex believes the combination of REQORSA® and Tecentriq can provide a promising new therapeutic option for the treatment of small cell lung cancer."
By Ryan Confer, President and Chief Executive Officer of Genprex.



Amidst the realms of cancer and diabetes treatment lies a daunting challenge: the limitations of conventional therapies in meeting the multifaceted needs of patients. Lung cancer patients often confront restricted options, grappling with drug resistance to common medications like Tagrisso®, Keytruda®, and Tecentriq®.
Simultaneously, the diabetes crisis persists, with millions enduring compromised quality of life due to outdated treatment methods, unchanged since insulin's inception in 1922. This glaring disparity underscores the urgent need for effective, sustainable solutions— a void that Genprex endeavors to bridge through its groundbreaking gene therapies.
Genprex’s unwavering dedication to reshaping patient care via innovative technologies serves as a glimmer of hope for those impacted by these debilitating illnesses. By addressing this widespread challenge head-on, the Company heralds a new epoch of transformative healthcare, promising renewed hope and improved outcomes for patients worldwide.

Genprex leads the charge in transformative healthcare, providing pioneering solutions to the prevalent obstacles in cancer and diabetes treatment. Through the Company’s cutting-edge non-viral gene therapy platform, a revolutionary method to combat lung cancer by systematically replacing tumor suppressor genes. REQORSA®, the Company’s flagship product candidate, symbolizes a beacon of optimism, showcasing promising results in clinical trials, including instances of tumor regression and enhanced patient responses.
In the realm of diabetes, Genprex’s gene therapy contenders, GPX-002, exhibit unparalleled potential. Designed to tackle both Type 1 and Type 2 diabetes, this candidate, delivered via an innovative infusion process, strive to restore normal blood glucose levels for extended durations. With a mission to redefine the landscape of cancer and diabetes treatment, Genprex is dedicated to propelling this groundbreaking gene therapies forward, bridging the divide in conventional therapies and steering towards a future where patients can embrace life with revitalized hope and heightened quality.



Genprex’s technologies are designed to administer disease-fighting genes to provide new therapies for large patient populations with cancer and diabetes who currently have limited treatment options. The Company’s teams work closely with world-class researchers, institutions and industry collaborators to expand Genprex’s pipeline of gene therapies and provide a wider range of novel treatment approaches for patients with cancer and diabetes.



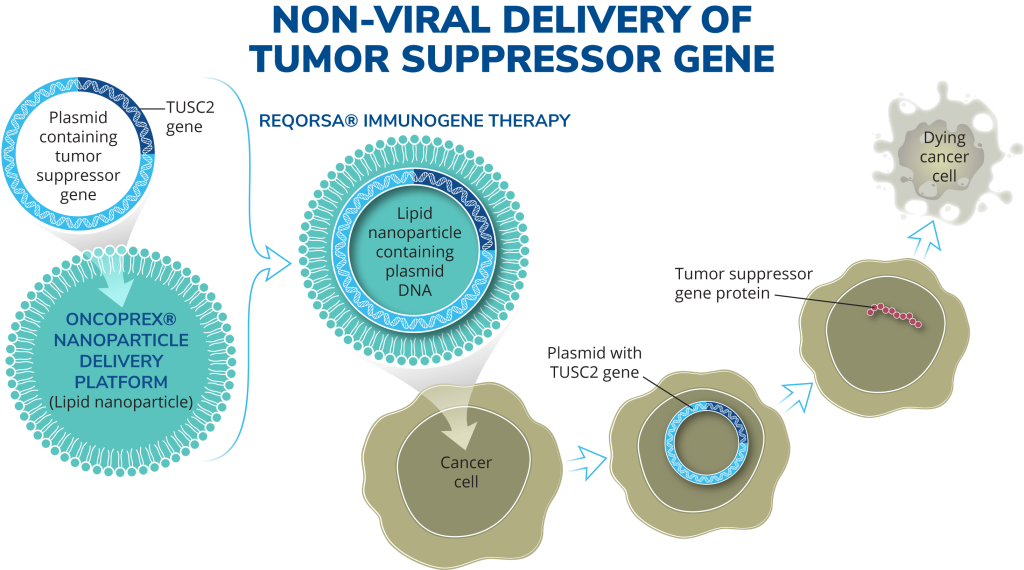
Genprex’s oncology drug development program utilizes its unique, proprietary, non-viral ONCOPREX® Nanoparticle Delivery System, which is believed to be the first systemic gene therapy delivery platform used for cancer in humans. Using this system, the company encapsulate plasmids that express tumor suppressor genes within lipid nanoparticles and intravenously administer the encapsulated plasmids which are taken up by the tumor cells, after which the tumor suppressor genes express proteins that are missing or found in low quantities in the tumor cells.
REQORSA® Immunogene Therapy, Genprex's flagship product candidate for non-small cell lung cancer (NSCLC) and small cell lung cancer (SCLC), utilizes the innovative ONCOPREX® Nanoparticle Delivery System, delivering the TUSC2 tumor suppressor gene. This therapy aims to disrupt cancer cell signaling pathways, induce cancer cell death, and enhance the immune response against cancer, serving as a blend of gene therapy and immunotherapy. Notably, REQORSA® received Fast Track Designations from the FDA in 2020 and 2021 for its combinations with AstraZeneca's Tagrisso® and Merck & Co's Keytruda®, respectively, and in 2023 for its combination with Genentech, Inc.'s Tecentriq® in extensive-stage small cell lung cancer (ES-SCLC) patients.

Administered intravenously, REQORSA® non-viral nanoparticles target cancer cells specifically, delivering the TUSC2 gene while minimizing uptake by normal tissues. Tumor biopsy studies revealed significantly higher uptake of TUSC2 in tumor cells compared to normal cells, demonstrating REQORSA® targeted delivery efficacy.
Unlike many gene therapies requiring permanent DNA changes, REQORSA® does not integrate into the patient's DNA, simplifying administration.
REQORSA® boasts a multimodal mechanism of action, disrupting cancer cell replication and survival pathways, inducing apoptosis, and modulating the immune response. This versatility extends to combination therapies, where REQORSA® has shown promising results in preclinical and clinical studies, potentially offering effective treatment options for a wide range of lung cancer patients.
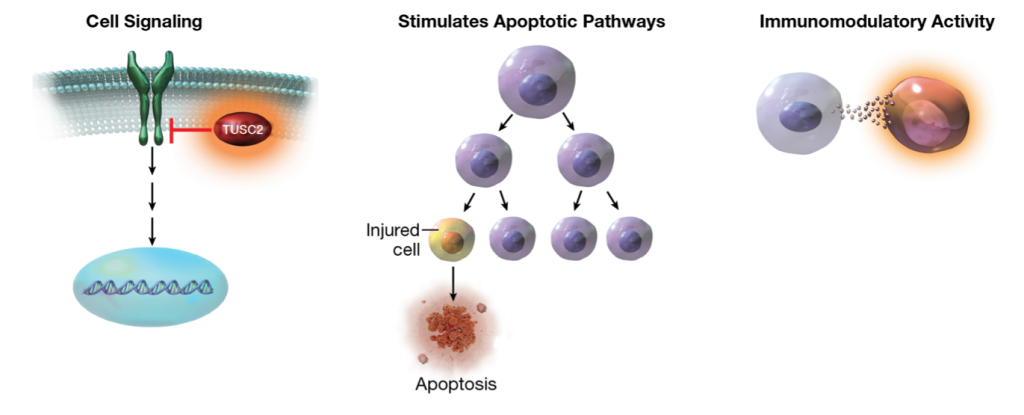

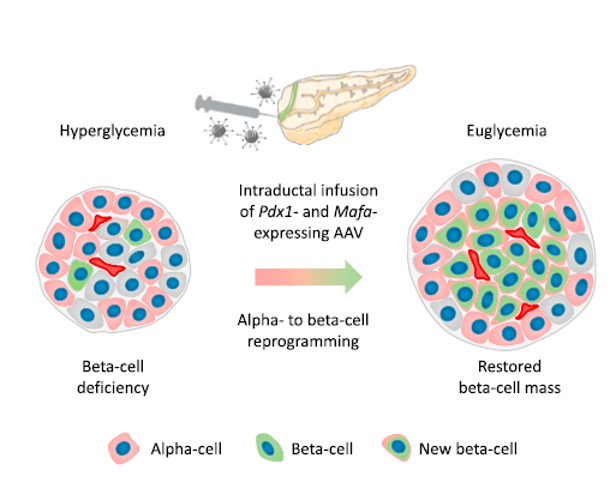
Genprex's diabetes gene therapy program introduces GPX-002, a groundbreaking solution developed in collaboration with the University of Pittsburgh. This therapy employs a novel infusion method using an adeno-associated virus (AAV) vector to deliver Pdx1 and MafA genes directly to the pancreas. In Type 1 diabetes models, these genes prompt alpha cells to transform into functional beta-like cells, capable of producing insulin while evading immune detection.
The innovative approach of Genprex's gene therapies extends to both Type 1 and Type 2 diabetes, utilizing the same AAV vector technology for targeted delivery. Preclinical studies conducted at the University of Pittsburgh are optimizing these therapies, paving the way for future clinical trials. GPX-002's efficacy in restoring normoglycemia and increasing insulin-producing cell mass has shown promising results in diabetic mouse models and nonhuman primates.
Recent presentations of preclinical data at international conferences underscore the therapeutic potential of GPX-002. Statistically significant outcomes in nonhuman primates include decreased insulin requirements, increased c-peptide levels, and improved glucose tolerance. These findings suggest a transformative breakthrough in diabetes treatment, with the potential to eliminate the need for insulin replacement therapy in both Type 1 and Type 2 diabetic patients.
Genprex's ONCOPREX® Nanoparticle Delivery System marks a pioneering advancement in oncology, revolutionizing the delivery of anti-cancer genes. Developed through collaborative research with esteemed institutions like the University of Texas MD Anderson Cancer Center and the National Institutes of Health, this non-viral platform delivers DNA plasmids containing cancer-fighting genes encapsulated in lipid nanoparticles intravenously to patients.

The ONCOPREX® system is optimized to deliver therapeutic payloads systemically, targeting cancer cells with precision. Utilizing positively charged nanoparticles, it selectively binds to negatively charged cancer cells and enters them through selective endocytosis. This unique approach ensures efficient delivery of therapeutic genes while minimizing uptake by normal cells.
Key to the efficacy of Genprex's delivery system is the optimized particle size, designed to navigate tight barriers in the lung while avoiding accumulation in vital organs like the liver, spleen, and kidney. Phase 1 clinical trials have demonstrated selective targeting of tumor cells, resulting in significant anticancer activity with minimal immunogenicity, allowing for repetitive therapeutic dosing.
With over 50 patients enrolled in Phase 1 and 2 clinical trials, Genprex has established the safety and efficacy of REQORSA® therapy using the ONCOPREX® Nanoparticle Delivery System. The therapy has shown promising results in selectively targeting tumor cells, demonstrating clinically significant anticancer activity while being well tolerated in humans. This groundbreaking approach represents a significant milestone in systemic gene therapy, offering new hope in the fight against cancer.
"Genprex is developing a robust research program to expand the potential tumor targets, and even non-tumor targets, that may be included in future clinical trials for REQORSA®. Research indicates that the TUSC2 gene used in REQORSA® may benefit many types of cancers and potentially the treatment of other diseases. The Company is exploring opportunities to treat other cancers in which TUSC2 is often deleted or inactivated, and is evaluating TUSC2 basic biology to better understand how to use REQORSA® treatment. Additionally, Genprex is exploring the use of another tumor suppressor gene, NPRL2, in cancer treatment using the ONCOPREX® Nanoparticle Delivery System."
By Rodney Varner, the late Chairman, President and Chief Executive Officer at Genprex.
Genprex's oncology program adopts a cutting-edge combination treatment approach, merging REQORSA® immunogene therapy with the company’s proprietary ONCOPREX® nanoparticle delivery system alongside approved targeted therapies and immunotherapies. This strategy offers renewed hope to large patient populations ineligible for these therapies or those who develop resistance.
Research demonstrates the synergistic effect of REQORSA® when combined with targeted therapies like Tarceva or Tagrisso, and immunotherapies such as Opdivo or Keytruda. By leveraging this synergy, Genprex aims to extend the benefits of these drugs to patients previously ineligible or resistant due to molecular profiles or drug tolerance.
Genprex's combination trials pave the way for expanding treatment options, addressing the limitations of targeted therapies which require specific genetic mutations. In January 2020, FDA granted Fast Track Designation for REQORSA® in combination with Tagrisso for NSCLC patients with EFGR mutations post-progression.
Additionally, Genprex's preclinical studies explore REQORSA® potential in combination with checkpoint inhibitors like Keytruda, indicating promising synergistic effects. This innovative approach holds promise for enhancing treatment efficacy beyond conventional therapies, ushering in a new era in cancer care.



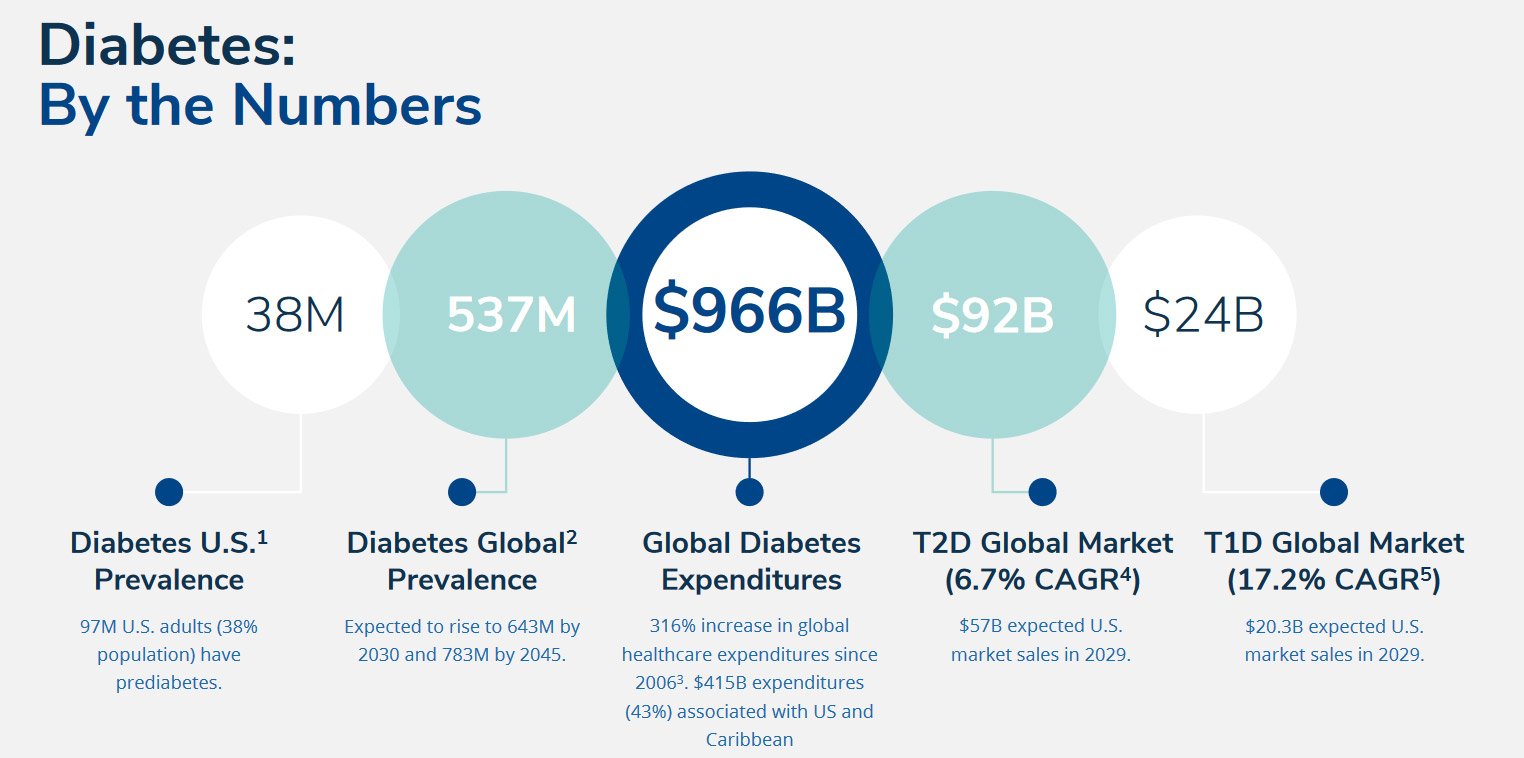
Cancer is a multifaceted illness characterized by uncontrolled cell growth, disrupting normal bodily functions. Lung cancer, Genprex's primary focus, encompasses non-small cell lung cancer (NSCLC) and small cell lung cancer (SCLC). According to the World Health Organization, lung cancer is the foremost cause of cancer-related deaths globally, surpassing colorectal, breast, liver, and stomach cancers in mortality rates.
In 2020, over 2 million new cases of lung cancer were reported worldwide, with 1.8 million resulting in fatalities. In the United States alone, projections by the American Cancer Society estimate more than 236,000 new lung cancer cases and over 130,000 deaths in 2022. NSCLC comprises 84% of lung cancer cases, with a five-year survival rate of merely 7% for those with distant metastasis. SCLC accounts for around 13% of lung cancer cases, with a dismal five-year survival rate of 3% in cases with distant spread.
Given the limited efficacy of existing therapies, there exists a substantial unmet medical need for innovative treatments for both NSCLC and SCLC globally. Genprex believes its lead candidate, REQORSA®, holds promise as a viable option for the majority of lung cancer patients, addressing this critical gap in care.

"Genprex is encouraged by the very promising Phase 1 results in the Acclaim-1 trial, which demonstrated that the combination of REQORSA® and Tagrisso was well tolerated at all three dose levels, with evidence of efficacy observed in patients with non-small cell lung cancer (NSCLC) whose disease has progressed on Tagrisso. Genprex is excited to begin the Phase 2a expansion portion of Acclaim-1, which will examine the toxicity profiles of different cohorts as well as efficacy and other clinical endpoints."
By Mark Berger, Chief Medical Officer of Genprex.



Genprex's diabetes gene therapy candidate, GPX-002, is being developed for the treatment of diabetes. According to the U.S. Center for Disease Control, 38.4 million Americans, or approximately 11.6% of the population, have diabetes. It is also believed that more than 96 million Americans have prediabetes, which represents approximately 38% of the U.S. population. The prevalence of this chronic disease is continuing to rise.
Chronic diabetes conditions include Type 1 diabetes and Type 2 diabetes, both of which lead to excess sugar in the blood and can cause serious health problems. Left untreated, high blood sugar levels can damage eyes, kidneys, nerves, and the heart, and can also lead to coma and death.
Genprex pioneers cutting-edge technologies, aiming to revolutionize cancer and diabetes treatments. REQORSA®, Genprex's lead product for lung cancer, is explored in tandem with targeted therapies and immunotherapies. Ongoing research extends the application to diverse tumors. The company's non-viral ONCOPREX platform underpins these advancements. In diabetes, GPX-002 promises breakthroughs. Genprex's dynamic pipeline signifies the Company’s commitment to transforming patient care with innovative gene therapies.
Revolutionizing Healthcare: Genprex stands at the forefront of transformative healthcare, pioneering gene therapies that redefine the treatment landscape for cancer and diabetes. Leveraging cutting-edge technologies and innovative science, Genprex's are committed to addressing the prevalent challenges in traditional therapies, offering groundbreaking solutions to patients worldwide.
Unmet Medical Needs: In the realm of cancer and diabetes treatment, significant unmet medical needs persist, affecting millions globally. With lung cancer ranking as the leading cause of cancer deaths and diabetes prevalence on the rise, there is a pressing demand for novel treatments that can provide hope and improve patient outcomes.
Breakthrough Therapies: Genprex's lead product candidate, REQORSA® Immunogene Therapy, represents a paradigm shift in cancer care, demonstrating promising outcomes in clinical trials. By leveraging the company proprietary ONCOPREX® Nanoparticle Delivery System, REQORSA® offers a targeted approach to combatting cancer, potentially reversing tumor growth and enhancing patient responses.
Expanding Opportunities: In addition to Genprex oncology pipeline, Genprex is spearheading innovative gene therapies for diabetes, addressing both Type 1 and Type 2 diabetes with Genprex GPX-002. This therapy offers hope to millions by targeting the root causes of the disease and restoring normal function to pancreatic cells.
Investment Potential: With upcoming milestones in clinical trials, promising preclinical data, and strategic partnerships, Genprex presents an enticing investment opportunity in the rapidly evolving field of biotechnology. Join Genprex as the Company paves the way for groundbreaking treatments that have the potential to transform lives and generate significant value for shareholders.
PLEASE NOTE WELL: Compass Live Media are not registered as an Investment Advisor in any jurisdiction whatsoever.
Stocks profiled on Compass Live Media web site are for informational purposes only. Information on these pages contains forward – looking statements that involve risk and uncertainties. The purpose of these profiles is to make investors aware of these companies and should not in any way come across as a recommendation to buy or sell in these securities. Investing in stocks involves risk. You should consult a qualified financial advisor or broker before making any investment decisions. All profiles are based on information that is available to the public. Past performance of stocks profiled on this web site is not a guarantee as to future performance. The information contained herein should not be considered to be all-inclusive and is not guaranteed by Compass Live Media to be free from misstatements or errors.
We do not give price targets in any of our written or recorded material.
Please do your own research and make your own investment decisions. Always remember that Compass Live Media is not an analyst and we do not employ or contract any analysts. Investing in securities such as the ones mentioned on our website, in email, or consulted for are for high-risk tolerant individuals only and not the general public.
Whether you are an experienced penny stocks investor or not, you should always consult with a licensed penny stock broker before buying or selling any securities that Compass Live Media profiles, mentions, in email updates, consults for or interviews. If shares are restricted, Compass Live Media may sell them when they are registered. If the penny stocks shares are freely transferable, Compass Live Media intends to sell them. At no time will any Compass Live Media employees or affiliates undertake any activity that could be regarded in any fashion as improper or illegal. It is possible that an investor’s investment may be lost or impaired due to the speculative nature of the companies profiled.
Many of the companies communicated in our emails or website are developmental stage companies with little or no operating or trading history. The information contained in our alerts should be viewed as commercial advertisement and is not intended to be investment opinion. The report is not provided to any particular individual with a view toward their investment circumstances. The information contained in our alerts is not an offer to buy or sell securities. We distribute opinions, comments and information free of charge to individuals who wish to receive them. Our advertisements and website have been prepared for informational purposes only and are not intended to be used as a complete source of information on any particular company. An individual should never invest in the securities of any of the companies profiles based solely on information contained in our report. Individuals should assume that all information contained in the alerts about profiled companies is not trust worthy unless verified by their own independent research. Any individual who chooses to invest in any securities should do so with caution. Investing in securities is speculative and carries a high degree of risk; you may lose some of all of the money that is invested. Always research your own investments and consult with a registered investment advisor or licensed stockbroker before investing. The profiles are a service of Compass Live Media, a marketing and advertising firm that may have been compensated. All direct and third party compensation received will be disclosed within each individual alert in accordance with section 17(b) of the nineteen thirty three Securities Act. Any compensation constitutes a conflict of interest as to our ability to remain objective in our communication regarding the profiled companies.
Compass Live Media distributes its Electronic Content and provides other promotional services with the intention of helping the “featured company and or companies” to create public awareness of their current business and future objectives, as well as to aid in the development of a liquid trading market for the “featured company’s” securities, although there is no intent to make a market or facilitate, in any manner, the purchase and sale of the “featured company and or companies” securities. Electronic Content refers to all Electronic Material distributed and created, and/or edited by Compass Live Media and includes, but is not limited to Emails, Newsletters, any Social Media content such as Facebook and Twitter, Blog Postings, Video Content, Corporate Profiles, Corporate Videos, Analyst Reports, PowerPoint Presentations, CEO Video Interviews, Press releases, Banners, Images, Google Advertising, Microsoft Ad center Advertising, and/or web based discussion board postings of any kind.
Although there is no guarantee that a liquid market will develop in the “featured company’s” securities, in the event that an active market does develop, investors should be aware that existing shareholders (controlling, or non-controlling) will be motivated to sell their shares potentially creating an oversupply scenario, under which the price of the shares is likely to experience sudden declines. At the same time, if the existing shareholder base holds shares tightly, and an active trading market develops, this may result in sudden increases in the price of the stock due to a potential undersupply scenario. In any case, there can be no guarantee that the price of the stock will reflect the fundamental value of the “featured company and or companies”. Compass Live Media disclaims all knowledge of the “featured company and/or companies” shareholder base, capacity for liquidity, or other means or methods of enhancing public awareness.
Forward Looking Statements
All forward-looking statements in this press release are expressly qualified by such cautionary statements and by reference to the underlying assumptions. None of the materials or advertisements herein constitute offers or solicitations to purchase or sell penny stocks securities of the penny stocks companies profiled herein and any decision to invest in any such penny stocks company or other financial decisions should not be made based upon the information provide herein. Instead Compass Live Media strongly urges you to conduct a complete due diligence of the respective penny stocks companies and consideration of all pertinent risks. Readers are advised to review SEC periodic reports: Forms 10-Q, 10K, Form 8-K, insider reports, Forms 3, 4, 5 Schedule 13D. Compass Live Media does not offer such advice or analysis, and Compass Live Media urges you to consult your own independent tax, business, financial and investment advisors. Investing in micro-cap and growth penny stocks securities is highly speculative and carries and extremely high degree of risk. It is possible that an investor’s investment may be lost or impaired due to the speculative nature of the companies profiled.
Compass Live Media may feature or profile a company periodically (usually for life of contract) and may remove that profile at it’s own discretion. Profiles and disclosures for those profiles may or may not remain visible on Compass Live Media website.
Compass Live Media website may contain links to related websites including, but not limited to, stock charts, stock quotes, SEC data, NASD data, etc. We are not responsible for the content of or the privacy practices used by these sites. We encourage our readers to review all public filings by companies at the SEC’s EDGAR page located at SEC.gov. The Compass Live Media has published information on how to invest carefully at NASD.com.
PLEASE READ CAREFULLY - Compass Live Media is engaged in the business of marketing and advertising companies for monetary compensation. All content in our releases is for informational purposes only and should not be construed as an offer or solicitation of an offer to buy or sell securities. Neither the information presented nor any statement or expression of opinion, or any other matter herein, directly or indirectly constitutes a solicitation of the purchase or sale of any securities. Compass Live Media sponsored advertisements do not purport to provide an analysis of any company’s financial position, operations or prospects and this is not to be construed as a recommendation by Compass Live Media or an offer or solicitation to buy or sell any security. Neither the owner of Compass Live Media nor any of its members, officers, directors, contractors or employees is licensed broker-dealers, account representatives, market makers, investment bankers, investment advisors, analyst or underwriters . Investing in securities, including the securities of those companies profiled or discussed on this website is for individuals tolerant of high risks. Viewers should always consult with a licensed securities professional before purchasing or selling any securities of companies profiled or discussed in our releases. It is possible that a viewer’s entire investment may be lost or impaired due to the speculative nature of the companies profiled. Remember, never invest in any security of a company profiled or discussed in a release or on our website unless you can afford to lose your entire investment. Also, investing in micro-cap securities is highly speculative and carries an extremely high degree of risk.
Notice of Stock Price Movements and Volatility Viewers of this newsletter should understand that trading activity and stock prices in many if not all cases tend to increase during the advertisement campaigns of the profiled companies and in many if not all cases tend to decrease thereafter. This tends to create above average volatility and price movements in the profiled company during the advertisement campaign that viewers should take into consideration at all times. Campaigns vary in length, and many are for short periods of time, typically less than a week. Some of the content in this release contains forward-looking information within the meaning of Section 27 A of the Securities Act of 1 993 and Section 21 E of the Securities Exchange Act of 1 934 including statements regarding expected continual growth of the profiled company and the value of its securities. In accordance with the safe harbor provisions of the Private Securities Litigation Reform Act of 1 995 it is hereby noted that statements contained herein that look forward in time which include everything other than historical information, involve risk and uncertainties that may affect a company’s actual results of operation. A company’s actual performance could greatly differ from those described in any forward looking statements or announcements mentioned in this release. Factors that should be considered that could cause actual results to differ include: the size and growth of the market for the company’s products; the company’s ability to fund its capital requirements in the near term and in the long term; pricing pressures; unforeseen and/or unexpected circumstances in happenings; etc. and the risk factors and other factors set forth in the company’s filings with the Securities and Exchange Commission. However, a company’s past performance does not guarantee future results. Generally, the information regarding a company profiled is provided from public sources which we believe to be reliable but is not guaranteed by us as being accurate. Further specific financial information, filings and disclosures as well as general investor information about the profiled company, advice to investors and other investor resources are available at the Securities and Exchange Commission (“SEC”) website www.sec.gov and the Financial Industry Regulatory Authority (“FINRA”) website at www.finra.org. Any investment should be made only after consulting with a qualified investment advisor and reviewing the publicly available financial statement and other information about the company profiled and verifying that the investment is appropriate and suitable.
Compass Live Media makes no representations, warranties or guarantees as to the accuracy or completeness of the information provided or discussed. Viewers should not rely solely on the information obtained in this release or on our website. Viewers should use the information provided by us regarding the profiled companies as a starting point for additional independent research on the companies profiled or discussed in order to allow the viewer to form his or her own opinion regarding investing in the securities of such companies. Factual statements, or the similar, made by the profiled companies are made as of the date stated and are subject to change without notice and Compass Live Media Network has no obligation to update any of the information provided. Compass Live Media, its owners, officers, directors, contractors and employees are not responsible for errors and omissions.
From time to time certain content in our releases or website is written and published by our employees or third parties. In addition to information about our profiled companies, from time to time, our releases and website will contain the symbols of companies and/or news feeds about companies that are not being profiled by us but are merely illustrative of certain activity in the micro cap or penny stock market that we are highlighting. Viewers are advised that all analysis reports and news feeds are issued solely for informational purposes. Any opinions expressed are subject to change without notice. It is also possible that one or more of the companies discussed or profiled in our release or on our website may not have approved certain or any statements within the website. Compass Live Media encourages viewers to supplement the information obtained from this release and our website with independent research and other professional advice. The content in this release is based on sources which we believe to be reliable but is not guaranteed by us as being accurate and does not purport to be a complete statement or summary of the available data.
This release may provide hyperlinks to third party websites or access to third party content. Compass Live Media its owners, officers, directors, contractors and employees are not responsible for errors and omissions nor does Compass Live Media control, endorse, or guarantee any content found in such sites. By accessing, viewing, or using the website or communications originating from this release, you agree that Compass Live Media its owners, officers, directors, contractors and employees, are not responsible for any content, associated links, resources, or services associated with a third party website. You further agree that Compass Live Media, its owners, officers, directors, contractors and employees shall not be liable for any loss or damage of any sort associated with your use of third party content. Links and access to these sites are provided for your convenience only.
Compass Live Media uses third parties to disseminate information to subscribers. Although we take precautions to prevent others from obtaining our subscriber list, there is a risk that our subscriber list, through no wrong doing on our part, could end up in the hands of an unauthorized party and that subscribers will receive communications from unauthorized third parties. You agree to hold Compass Live Media its operators, owners and employees harmless and to completely release them from any and all liability due to any and all loss (monetary or otherwise), damage (monetary or otherwise), or injury (monetary or otherwise) that you may incur arising out of the use of our website or the information in our press releases, profiles and opinions. You agree that use of our website is at your sole risk. Compass Live Media disclaims all warranties of any kind, express or implied. We encourage viewers to invest carefully and read the investor issuer information available at the web sites of the SEC. The SEC has launched an investor-focused website to help you invest wisely and avoid fraud at www.investor.gov and filings made by public companies can be viewed at www.sec.gov and/or then FINRA at: www.finra.org. In addition, FINRA has published information at its website on how to invest carefully at www.finra.org/investors/index.htm.
(Last updated: )